Équipez vos chambres. Protégez vos résidents.
Le matelas Decubi répond aux exigences des établissements de soin.
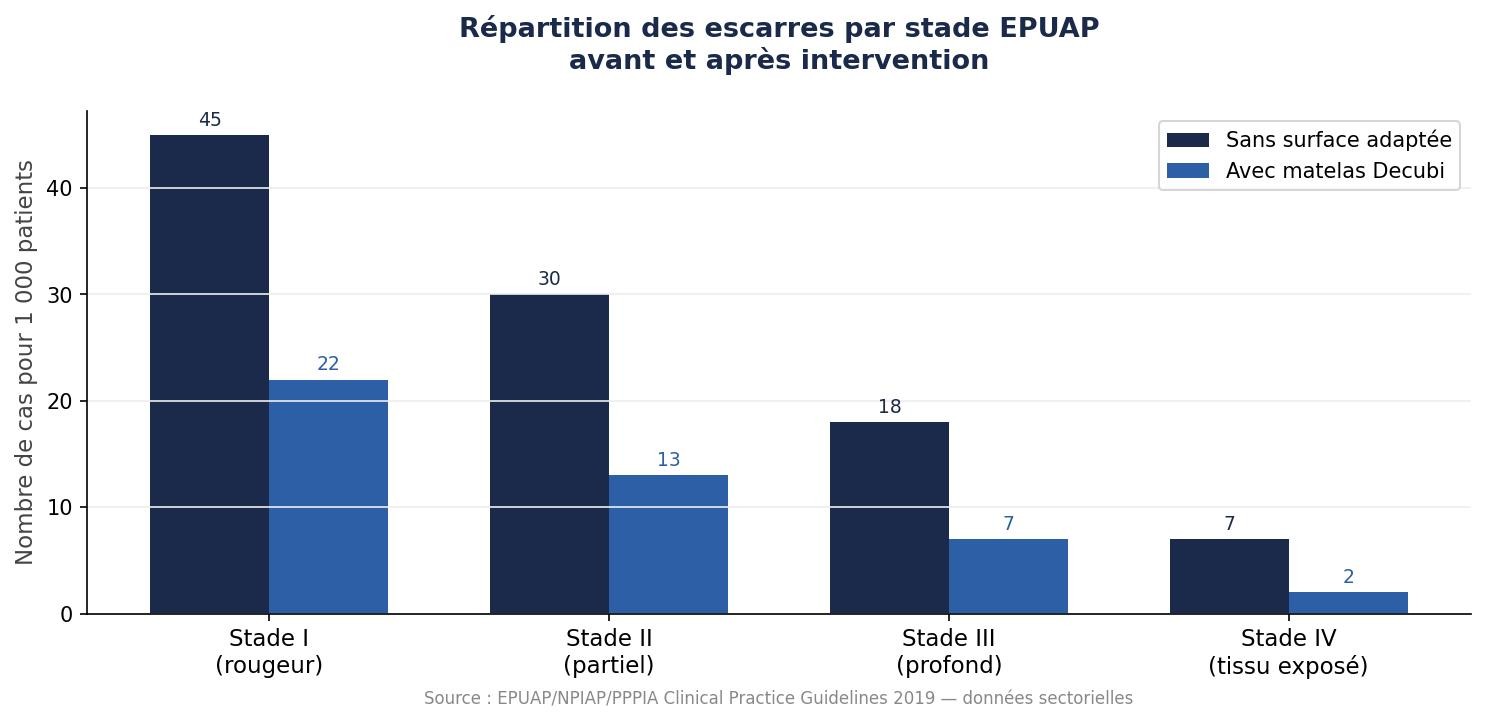
Certifié EPUAP stade III. Autoclavable 121°C. Compatible tous lits médicalisés.
Tarifs institutionnels sur devis à partir de 5 unités.
- Decubi : une solution cliniquement fondée pour la prévention des escarres.
Recommandé pour les patients à risque modéré à élevé selon la classification EPUAP. Compatible avec les protocoles aSSKINg et les supports de lit médicalisés. Sans alimentation électrique. Sans configuration.
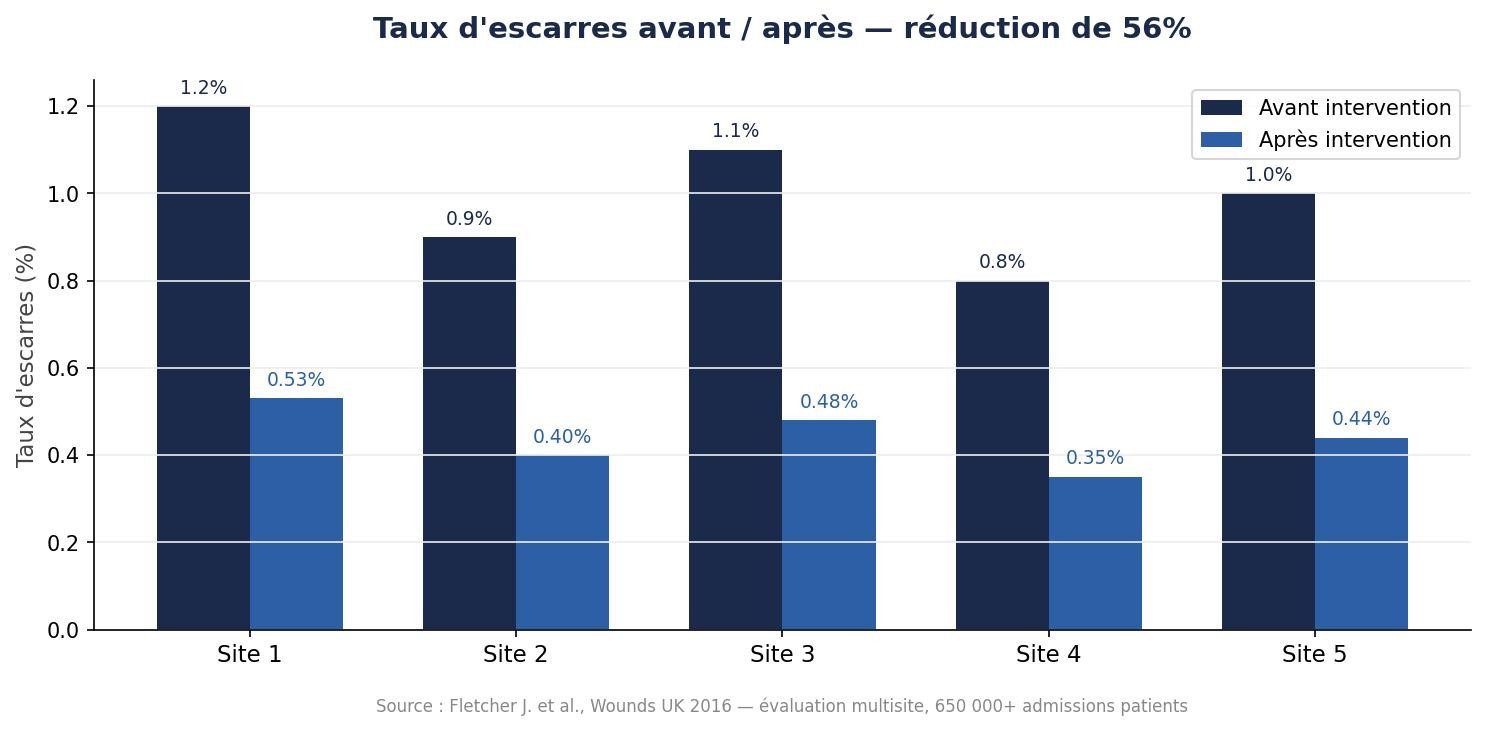
Réduction de 56% du taux d'escarres — évaluation multisite

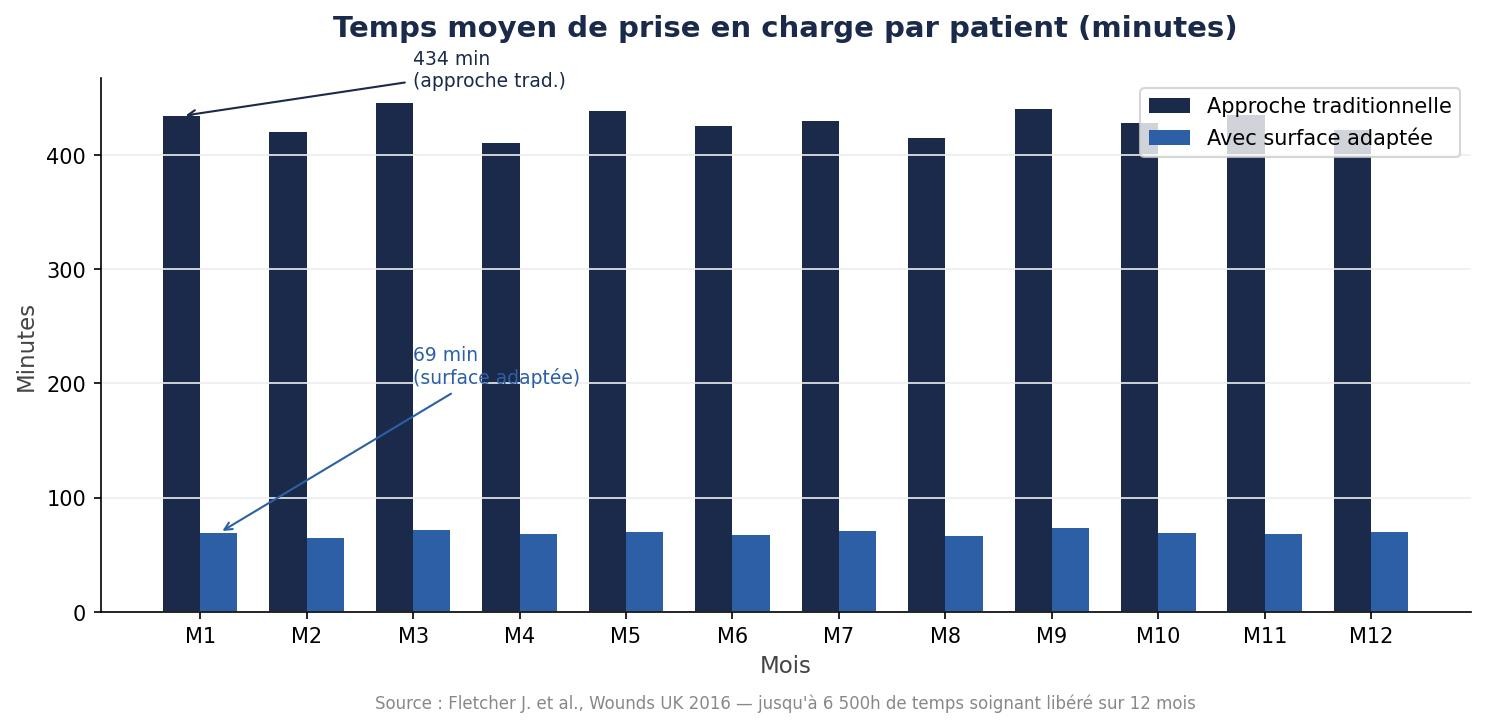
6 500h de temps soignant libéré en 12 mois
Certifications & garanties

L'impact économique d'une prévention efficace
Une escarre de stade III coûte entre 5 000€ et 15 000€ en soins (pansements, hospitalisations, personnel soignant supplémentaire).
Le matelas Decubi coûte 369€ l’unité (tarif dégressif dès 5 unités).
Le retour sur investissement est immédiat dès la prévention d’un seul cas.
Stat clé : 56% de réduction des escarres avec surface de support adaptée
(Source : DHG Healthcare / Wounds UK 2016)
Pourquoi les établissements choisissent le matelas Decubi ?
Une surface de support médicale passive, certifiée EPUAP, adaptée à tous les environnements de soins.
- Conforme aux protocoles aSSKINg et recommandations EPUAP 2019
- Housse autoclavable 121°C — conforme infection control
- Lavage machine 95°C — entretien simplifié pour le personnel
- Compatible 100% des lits médicalisés inclinables du marché
- Taille standard 90 × 200 cm — compatible la majorité des chambres
- Charge certifiée 150kg — adapté aux patients bariatriques léger
- Garantie 2 ans constructeur
- Tarifs dégressifs dès 5 unités

Obtenez votre devis institutionnel en 24h
Vous gérez un établissement de soins, une maison de repos ou un service de réhabilitation ?
Remplissez le formulaire ci-dessous et recevez une offre tarifaire personnalisée, une documentation clinique complète et un code de réduction dédié à votre structure.
Tarifs dégressifs à partir de 5 unités.
Pourquoi choisir le matelas Decubi ?



Ce que vous recevez
- Matelas Decubi — 90 × 200 cm — 14 cm de hauteur
- Housse PU OEKO-TEX® zippée — déjà montée, prête à l'emploi
- Emballage sous vide — décompresse en 4 à 8 heures après ouverture
- Guide d'utilisation & d'entretien — FR / NL / EN
- Certificat de conformité EPUAP
- Fiche technique produit
- Facture pour remboursement mutuelle
4.8 / 5
Ils ont choisi Decubi
Des familles, soignants et établissements qui font confiance à Decubi pour la prévention des escarres.
Questions fréquentes
Quelle est la taille du matelas ?
Le matelas Decubi est disponible en une seule taille : 90 × 200 cm (hauteur 14 cm).
Compatibilité : tous les cadres de lit médicalisés standard.
Puis-je le retourner si je ne suis pas satisfait ?
Une fois l’emballage sous vide ouvert, le retour n’est pas possible pour raisons d’hygiène
(exception légale art. VI.53 CDE belge). Vérifier la compatibilité avant d’ouvrir.
La mutuelle rembourse-t-elle ce matelas ?
Selon votre mutuelle. Nous fournissons une facture médicale détaillée pour votre demande.
Combien de temps pour reprendre sa forme ?
4 à 8 heures après ouverture de l’emballage sous vide. Poser à plat sur le lit dès réception.
Protégez vos clients dès aujourd'hui.
Le matelas Decubi — 90 × 200 cm — est disponible dès maintenant.
Livraison en Belgique et en Europe. Emballage sous vide. Facture pour la mutuelle.
